Eventually both objects will reach the same temperature, at a value between the initial temperatures of the two objects. If a hot frying pan that has just been removed from the stove is allowed to come into contact with a cooler object, such as cold water in a sink, heat will flow from the hotter object to the cooler one, in this case usually releasing steam. Let’s consider a familiar example of spontaneous change. This information, however, does not tell us whether a particular process or reaction will occur spontaneously. You also learned in Chapter 5 "Energy Changes in Chemical Reactions" that the enthalpy change for a chemical reaction can be calculated using tabulated values of enthalpies of formation. According to Section 18.2 "The First Law of Thermodynamics", changes in the internal energy (Δ E) are closely related to changes in the enthalpy (Δ H), which is a measure of the heat flow between a system and its surroundings at constant pressure. 
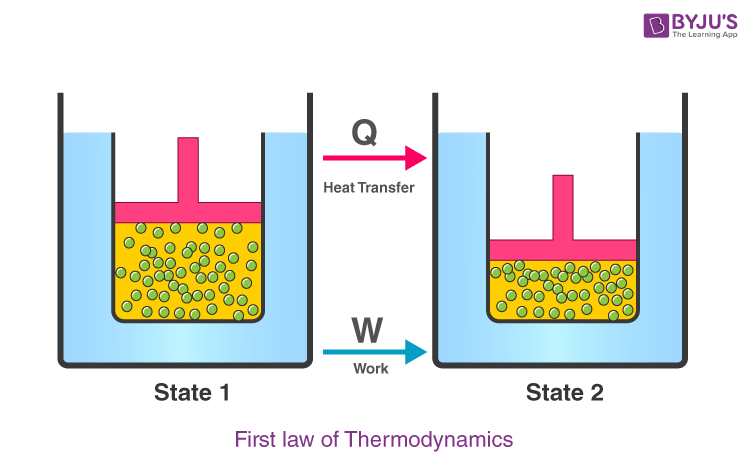
The first law of thermodynamics governs changes in the state function we have called internal energy ( E).  
To understand the relationship between internal energy and entropy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
